COLUMBIA | Zuckerman Institute
Polleux Lab
Circuit development, evolution and maintenance
Research
Our work provides new insights into the cellular and molecular mechanisms underlying the establishment and maintenance of brain connectivity and has significant implications for our understanding of the pathophysiological mechanisms underlying socially-devastating neurodevelopmental disorders and neurodegenerative diseases.
About the PI

Dr. Franck Polleux did his undergraduate and graduate studies at Université Claude Bernard in Lyon, (France) where he obtained his Ph.D. in Neuroscience in 1997. He then joined the laboratory of Dr. Anirvan Ghosh at Johns Hopkins University for his post-doctoral training.
In 2002, Dr Polleux was hired as an Assistant Professor in the Neuroscience Center and Department of Pharmacology at the University of North Carolina- Chapel Hill where he became an Associate Professor in 2008. In August 2010, he joined The Scripps Research Institute in La Jolla, California. In November 2013, he was recruited as a Professor in the Department of Neuroscience at Columbia University to join the new Mortimer B. Zuckerman Mind, Brain, Behavior Institute.
Throughout his career, Dr Polleux has focused on the identification of the molecular mechanisms underlying neuronal development in the mammalian brain. More recently, his lab started studying the genetic basis of human brain evolution as well as the signaling pathways underlying synaptic loss during early stages of Alzheimer’s Disease progression.
The Team

Daniel Virga (he/him)
Graduate Student

Stevie Hamilton (she/her)
Graduate Student

Kevin Gonzalez
Graduate Student

Sergio Bernal-Garcia (he/him)
Graduate Student

Victoria L. Hewitt (she/her)
PostDoc

Justin O'Hare
PostDoc

Eugenie Peze-Heidsieck (she/her)
PostDoc

Martina Proietti Onori (she/her)
PostDoc

Carlos Diaz Salazar Albeda
PostDoc

Qiaolian Liu
Research Technician

Rhythm Sharma (she/her)
Lab Manager
A sneak peak at some of our Tools
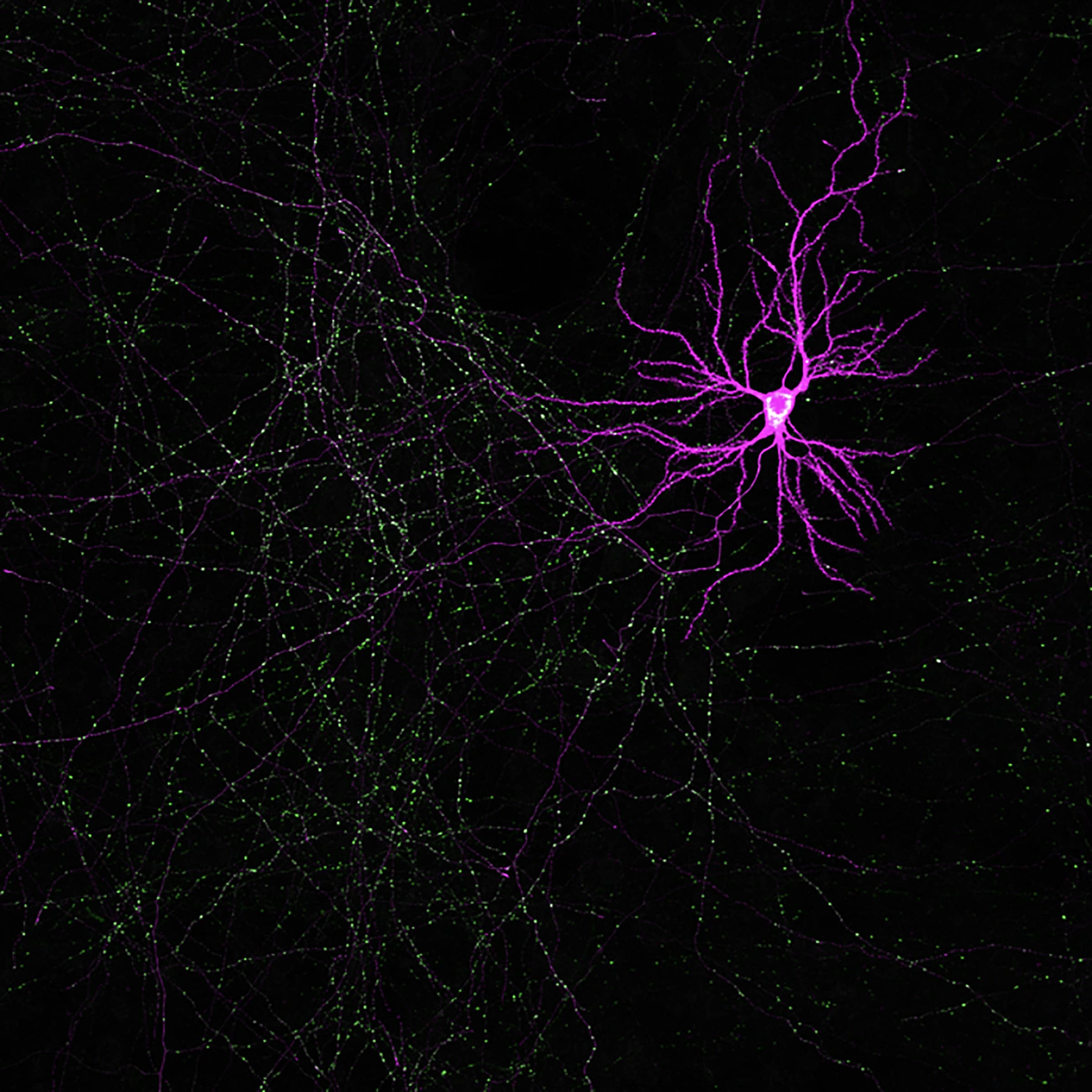
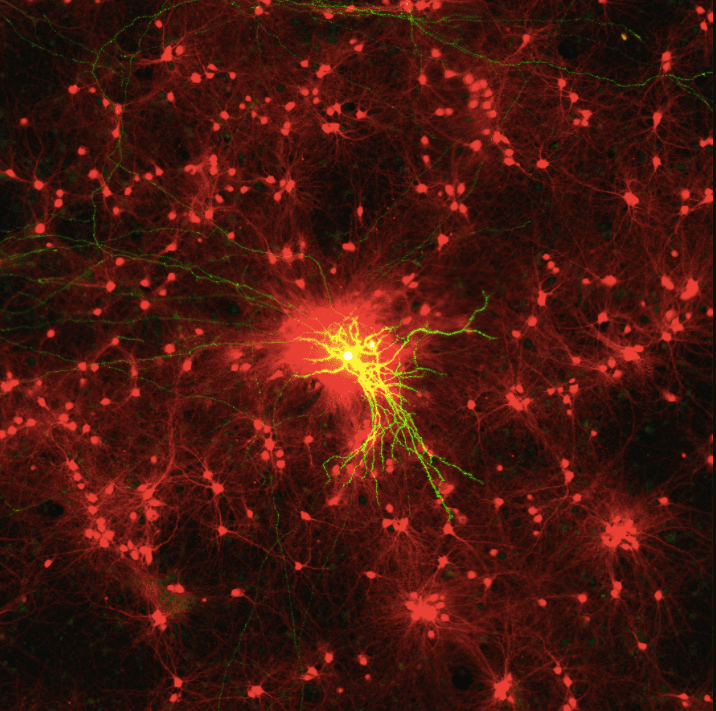
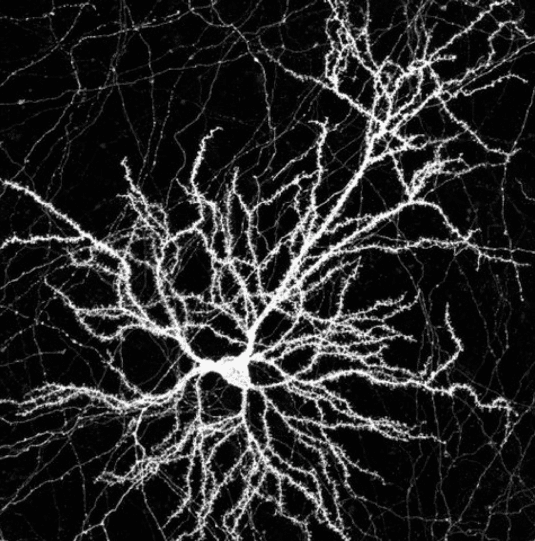
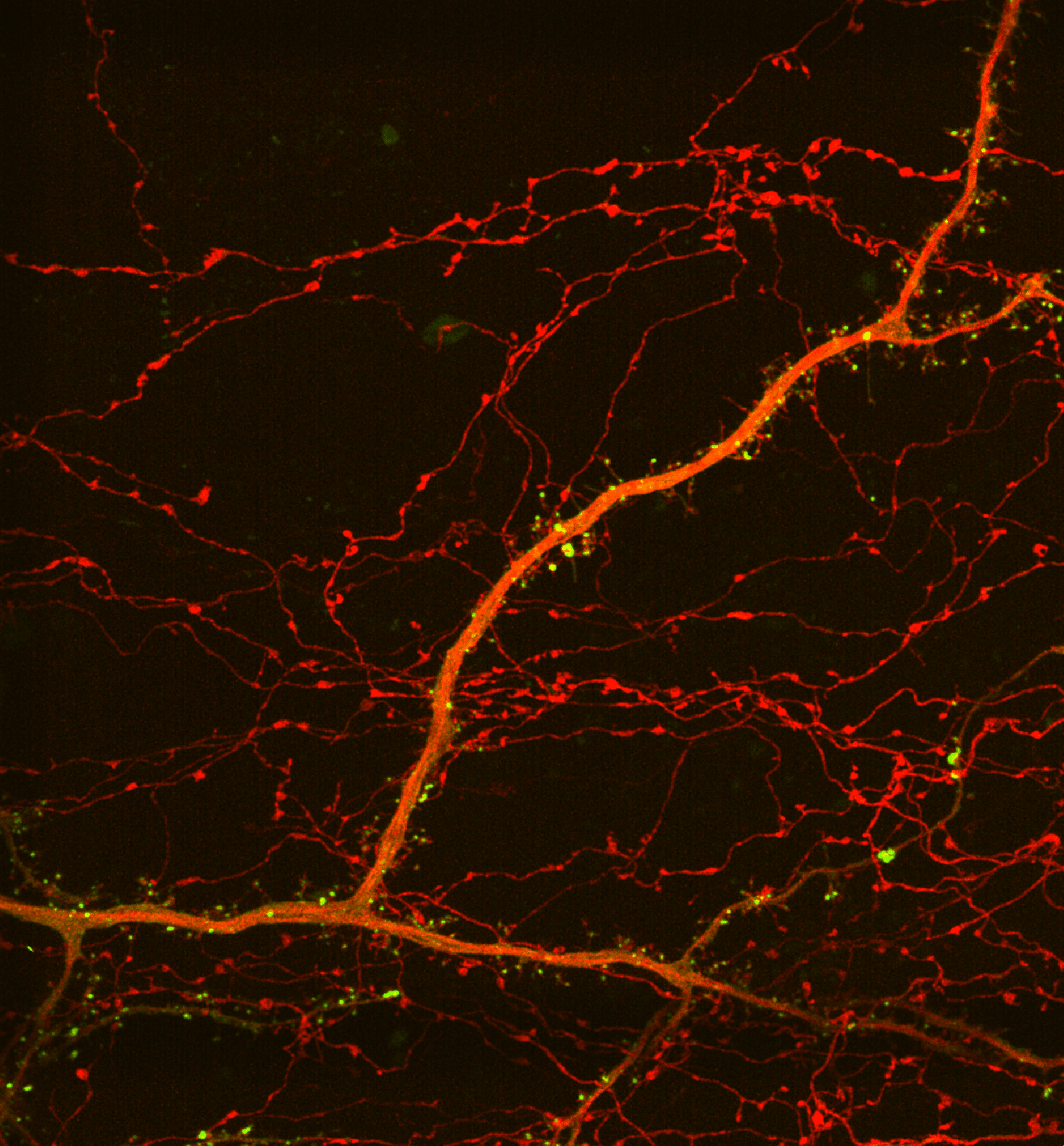
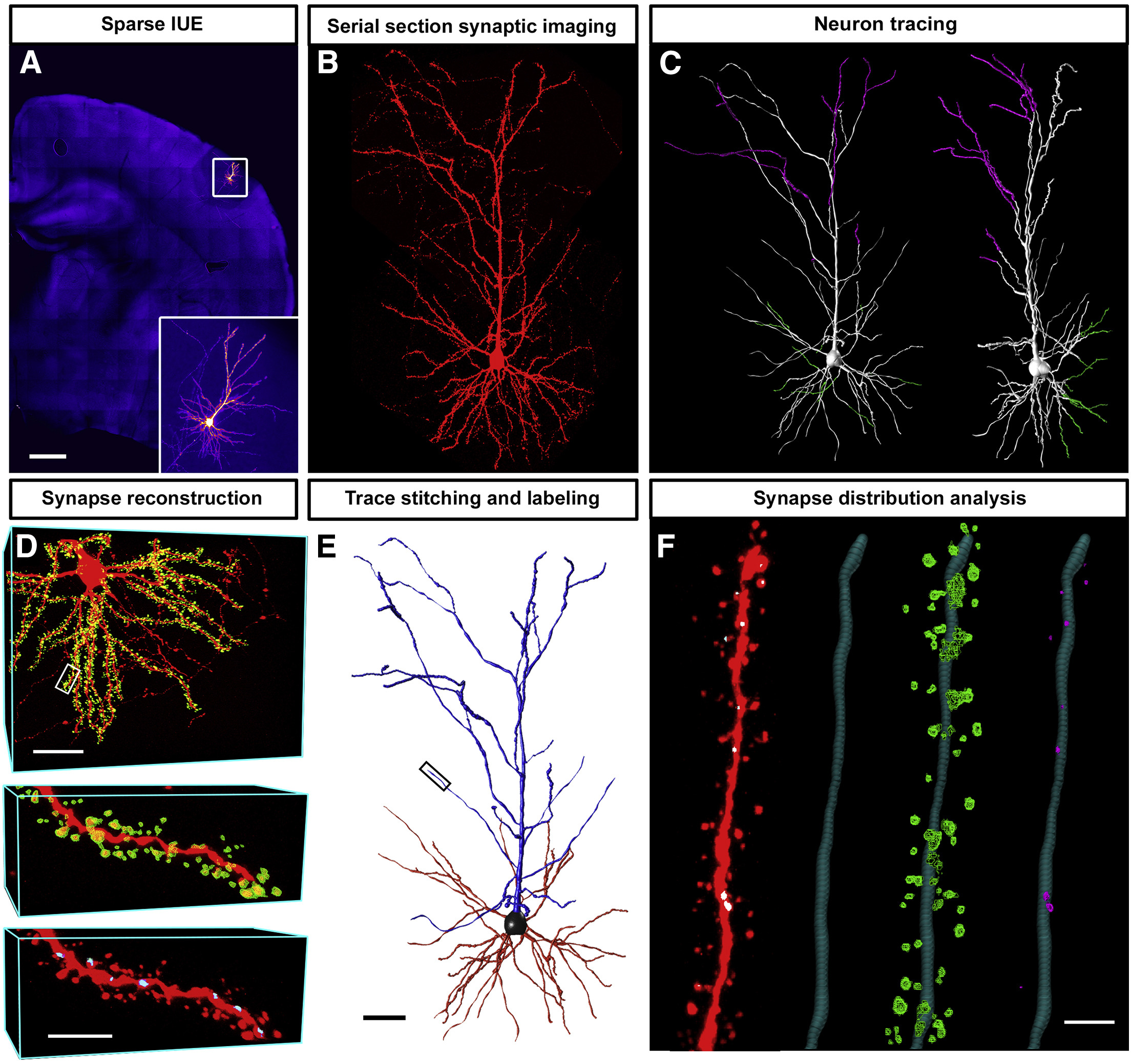
In Utero Electroporation and Neuron Reconstruction
The image describes the workflow for excitatory and inhibitory synaptic reconstruction across whole neurons with Vaa3D Synapse Detector program developed in collaboration with the Allen Institute for Brain Science.
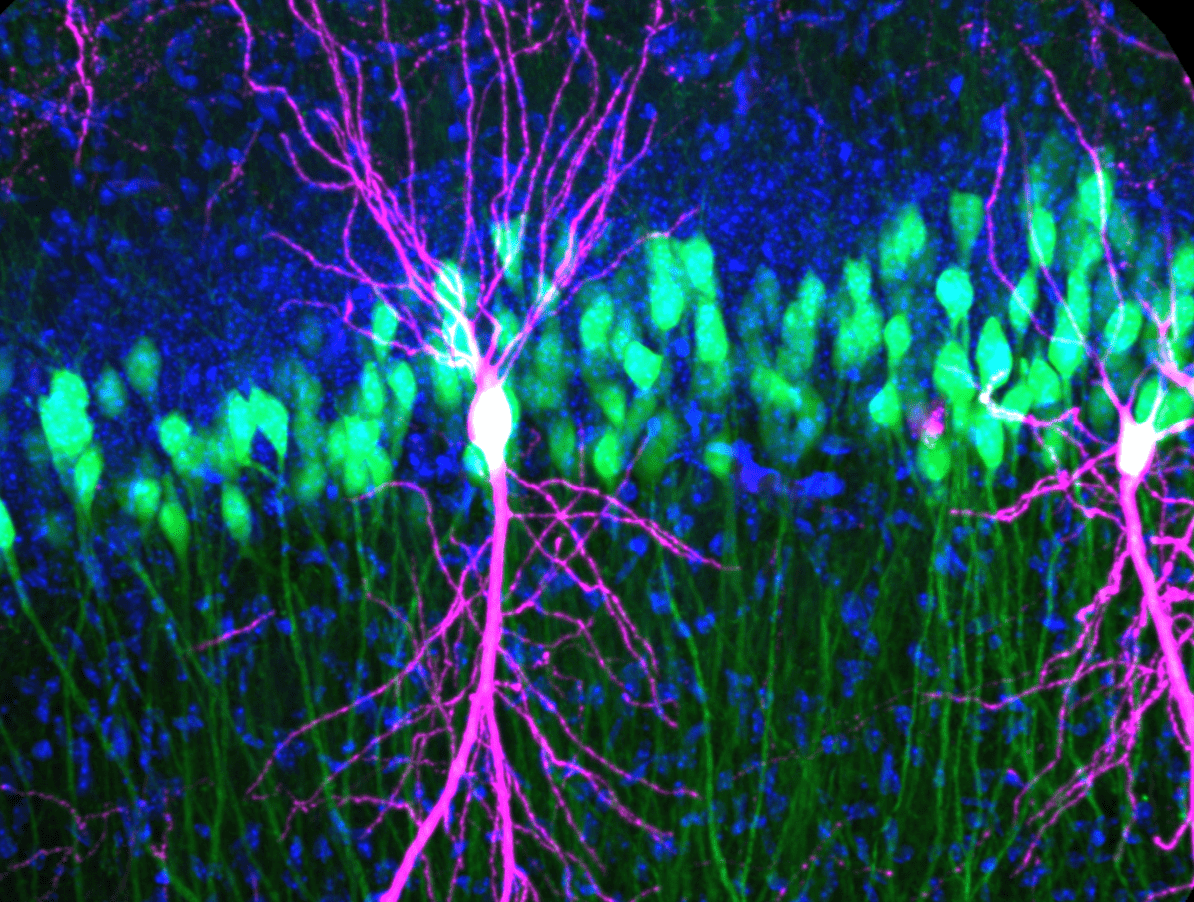
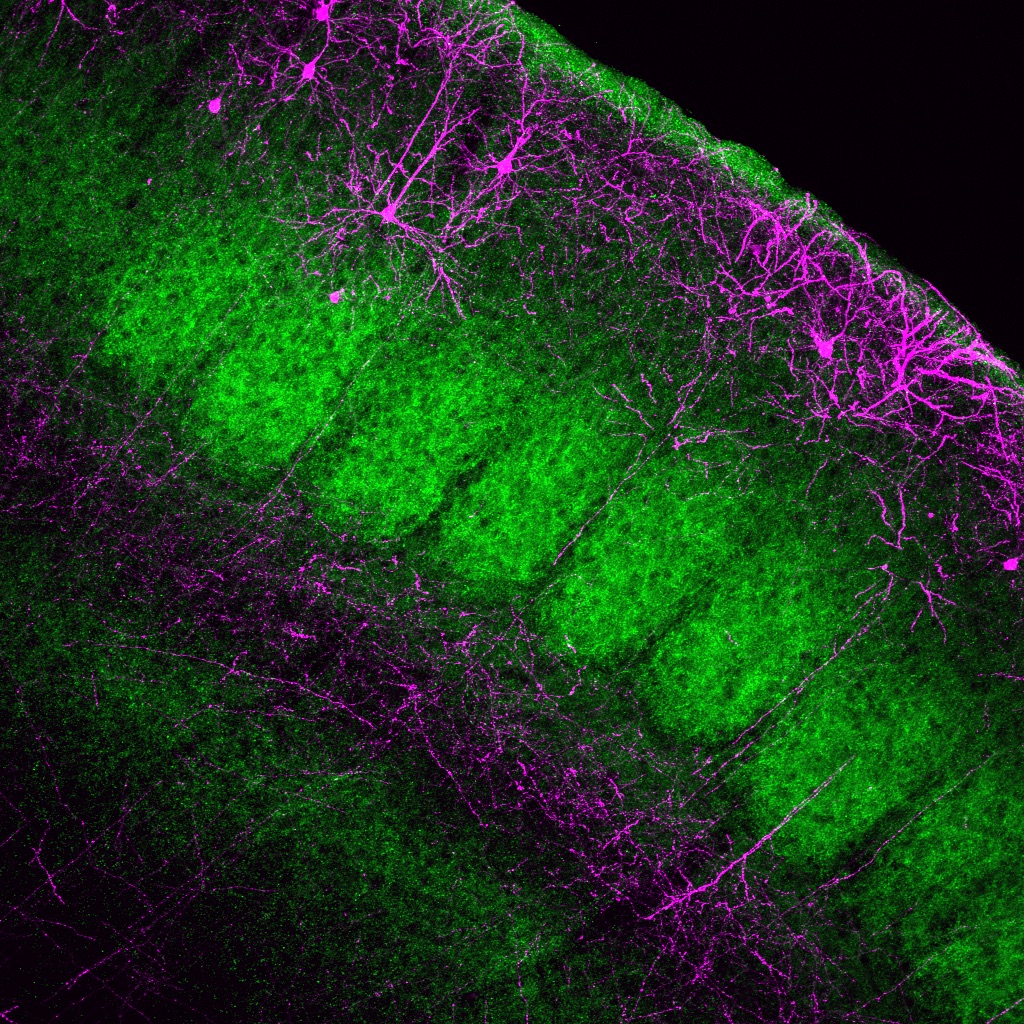
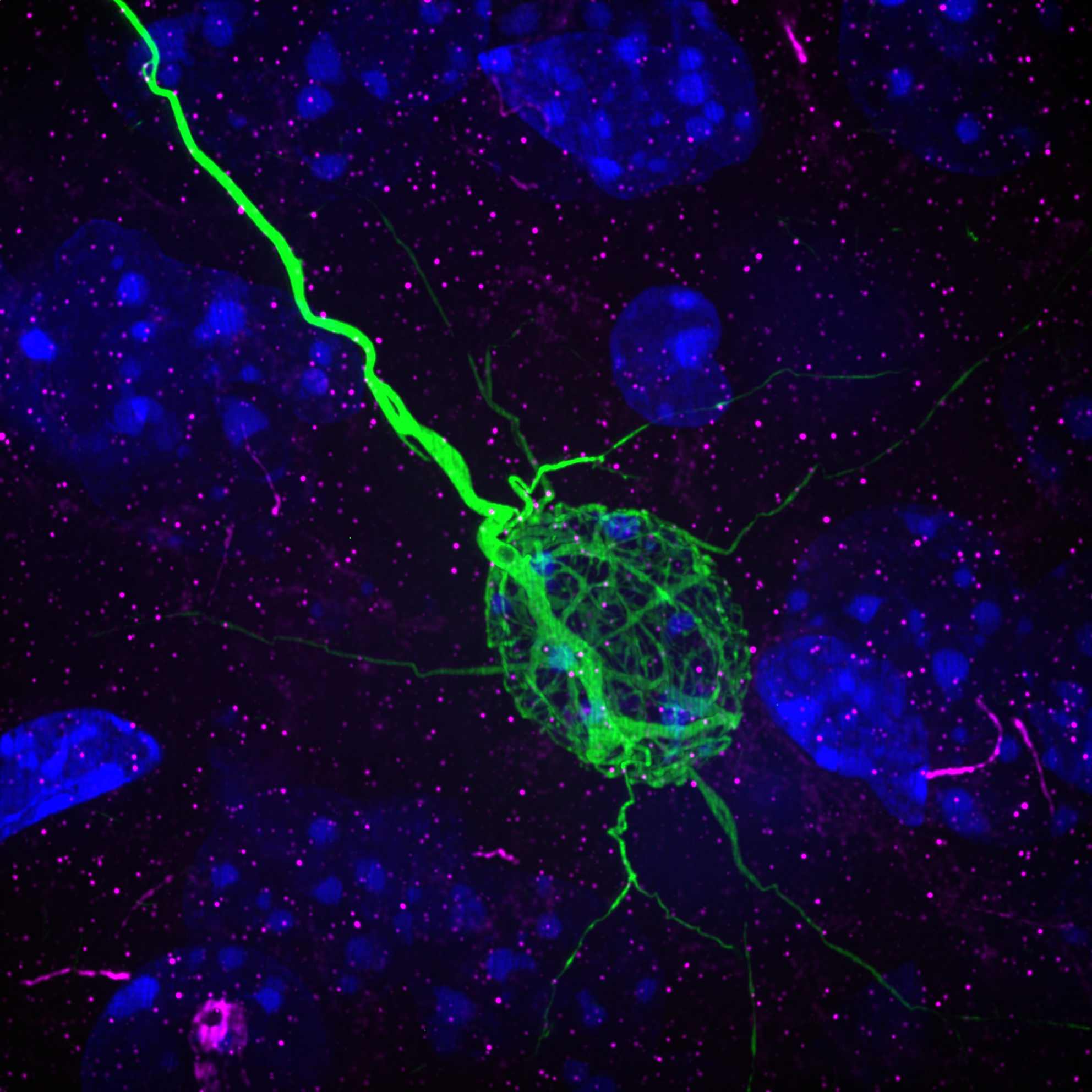
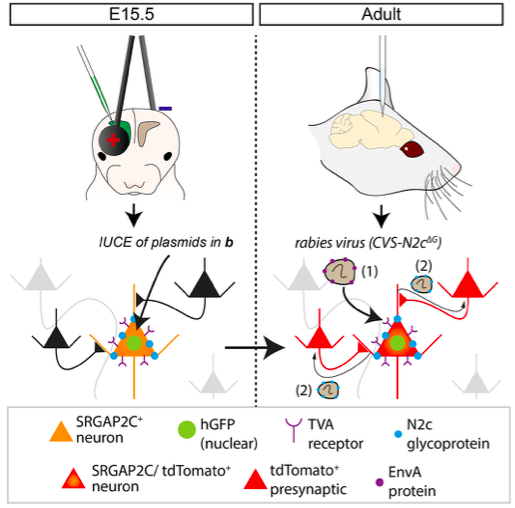
Rabies Tracing and Neural Connectivity Reconstruction
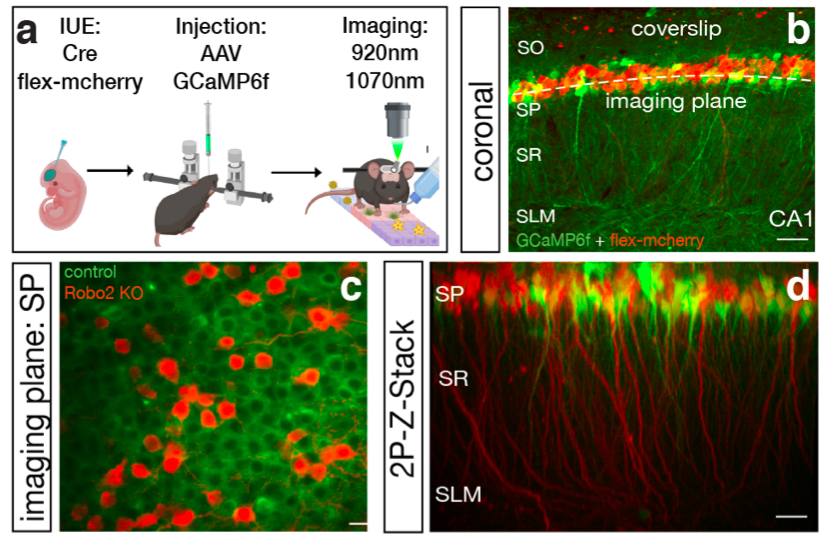
2-Photon Calcium Imaging in awake behaving mice
Publications and News
Funders




 )}
)}